Biocidal products are a broad group of chemicals that contain or generate active ingredients that give them antibacterial, antimicrobial, disinfecting or sanitising properties. They destroy, make harmless or control harmful organisms such as bacteria or viruses by means other than mere physical or mechanical action.
This information sheet covers biocidal products relevant to food safety.
Legislation
Regulation (EU) 528/2012 (The Biocidal Products Regulation or BPR) sets out the rules for the placing of Biocidal Products on the market and this is set out in Irish Law by S.I. 427/2013.
BPR defines biocides as follows:
- Any substance or mixture, in the form in which it is supplied to the consumer, consisting of, containing or generating one or more active substances, with the intention of destroying, deterring, rendering harmless, preventing the action of, or otherwise exerting a controlling effect on, any harmful organism by any means other than mere physical or mechanical action.
- Any substance or mixture, generated from substances or mixtures which do not themselves fall under the first indent, to be used with the intention of destroying, deterring, rendering harmless, preventing the action of, or otherwise exerting a controlling effect on, any harmful organism by any means other than mere physical or mechanical action.
- A **Treated Article that has a primary Biocidal function should be considered a Biocidal product
**Treated Article means any substance, mixture or article which has been treated with, or intentionally incorporates, one or more Biocidal products.
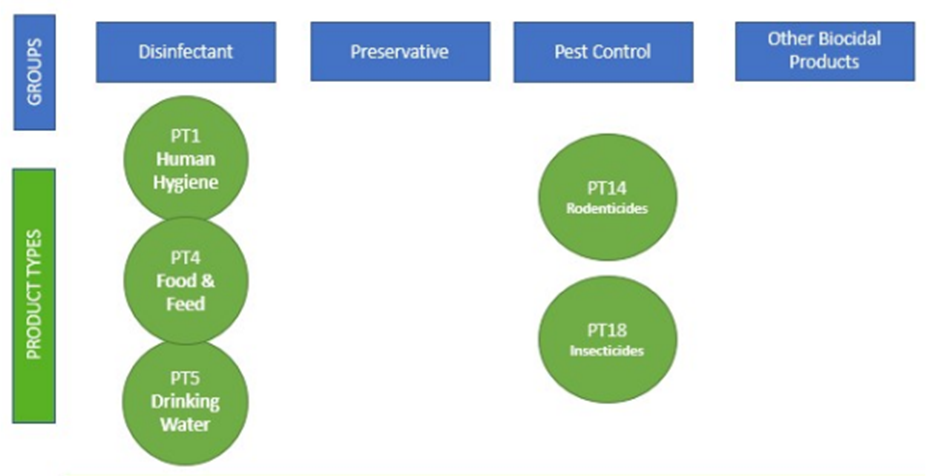
Under the BPR, biocidal actives are allocated to both Main Groups and Product Types (PTs). There are 4 Main Groups and 22 PTs, of which main groups 1 and 3 respectively are most relevant for food safety – see graphic
Regulation of Biocides in Ireland
The Department of Agriculture Food and the Marine (DAFM) is the central competent authority for the regulation of biocidal products in Ireland. Find detailed information on approval, labelling, register of notified/authorised products here: https://www.pcs.agriculture.gov.ie/biocides/biocidalproducts/
Notified and Authorised Products
Biocidal products cannot be placed on the market in Ireland unless they have been notified to/authorised by DAFM. Notified products will bear a number in the form of PCS XXXXXX whilst authorised products will bear a number in the form of IE/BPA 7XXXX or EU-000XXX-XX. They will also be included on the register of products.
Authorised products are those containing actives approved following a review process at EU level whereas notified products have yet to be reviewed. It will take several more years to complete this review process.
These links contain mock labels, mandatory label requirements and the Products Register:
- https://www.pcs.agriculture.gov.ie/biocides/biocidalproducts/packagingandlabelling/
- https://www.pcs.agriculture.gov.ie/registers/biocidalproductregisters/
Checklist for Food Inspectors
- Biocidal products will claim a biocidal effect such as antibacterial, antimicrobial, sanitiser, disinfectant etc.
- Does the product have a legitimate traceable source? (i.e. is the name and address of the registration holder/authorisation visible on the label)
- Does the product have a valid PCS or IE/BPA number on the label? This can be cross-checked on the DAFM products register.
- Is the product being used according to the label information? Remember that products with the same biocidal active substance but formulated for different purposes may not be safely interchangeable as they may contain different co-formulant chemicals which may be hazardous to food or operator health.
- For food contact surfaces, the product must be registered as Product Type 4 (PT 4).
- Does the label bear statutory details?
- biocidal active and its concentration.
- what the product is approved for. (e.g.is it approved as a PT 4 for use in food and feed areas?)
- type of product formulation.
- instructions for handling/usage/disposal
- Please note that DAFM have not approved any products for the control of COVID-19 through fogging/misting.
- Where unapproved products and/or product usage is encountered, the risk to food safety must be assessed and appropriate action taken. DAFM Biocides section should also be informed at biocide-notifications@agriculture.gov.ie.
- Products classified as hazardous must also be hazard labelled in accordance with Regulation (EC) No 1272/2008 on the Classification, Labelling and Packaging of Substances and Mixtures (CLP)
